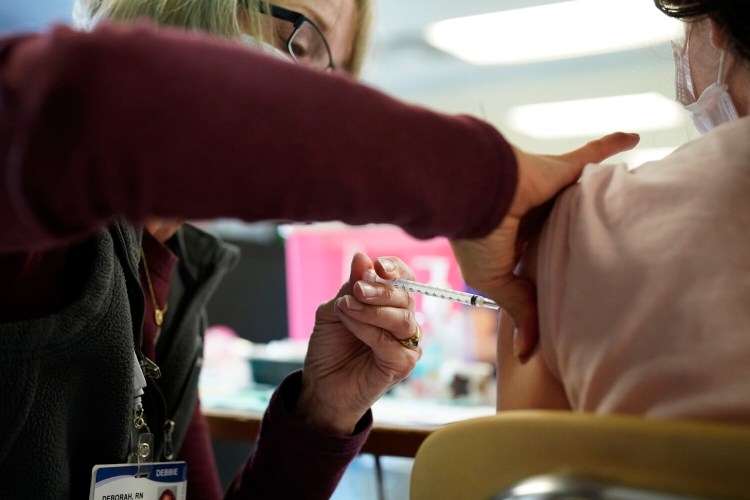
The Food and Drug Administration is expected by early next week to authorize booster shots of the Pfizer-BioNTech coronavirus vaccine for 12-to-15-year-olds, according to two people familiar with the FDA’s plan.
Some FDA officials had originally hoped to authorize a third dose of the Pfizer-BioNTech vaccine this week but had faced scheduling challenges with the holidays, according to the people with knowledge of the agency’s plan who spoke on the condition of anonymity to describe developing actions.
The FDA decision would then be reviewed by vaccine advisers at the Centers for Disease Control and Prevention, and that agency’s top official this week vowed to move quickly on recommending the booster shots if the advisers concurred with FDA.
“Of course, the CDC will swiftly follow as soon as we hear from them, and I’m hoping to have that in the days to weeks ahead,” CDC Director Rochelle Walensky said Wednesday on CNN.
Federal officials, public health experts and a growing number of parents have raised concerns that younger adolescents have been left disproportionately vulnerable to the fast-spreading omicron variant compared with other populations.
Older teenagers are already eligible for boosters, and younger children have more recently received their initial vaccine doses, which means they probably have retained more immune protection than children who were inoculated earlier.
The FDA’s plan was first reported by the New York Times.
FDA declined to comment.
Pfizer referred questions about booster shot timing to the FDA.
“As the booster is already authorized for 16 and over, we are confident regulators are making every effort to look for ways to preserve a high level of protection against the virus across broad populations,” Jerica Pitts, a Pfizer spokeswoman, wrote in an email.
Send questions/comments to the editors.


Success. Please wait for the page to reload. If the page does not reload within 5 seconds, please refresh the page.
Enter your email and password to access comments.
Hi, to comment on stories you must . This profile is in addition to your subscription and website login.
Already have a commenting profile? .
Invalid username/password.
Please check your email to confirm and complete your registration.
Only subscribers are eligible to post comments. Please subscribe or login first for digital access. Here’s why.
Use the form below to reset your password. When you've submitted your account email, we will send an email with a reset code.